Register here
What Is This San Diego Biotech Networking Event?
February 16, 2025 | San Diego
This San Diego biotech reception is an open, informal networking evening hosted by Crystal NAX, Crystal Pharmatech and Crystal Bio Solutions on February 24, 2026. It is designed for teams working on biologics, nucleic acids and small molecules.
From our side, we will bring experts in RNA therapeutics and LNP delivery. Our colleagues at Crystal Bio Solutions and Crystal Pharmatech will talk about biologics and small molecules respectively.
This event is intended for teams interested in:
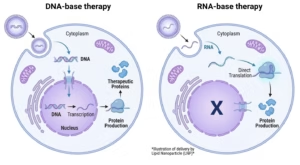
- Therapeutic RNA development: including projects in mRNA, saRNA, siRNA, ASO and gRNA.
- RNA manufacturing: design of impurity strategy, analytical readiness, and lifecycle planning
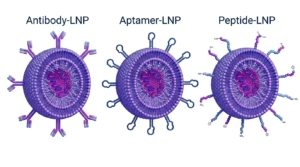
- LNP and delivery development: formulation, process development, and scale-up risk points
If you are evaluating how to control impurities, define release and stability strategies, or align analytics with your delivery approach, this reception supports early decision-making.
"The purpose is peer-level discussion about what actually de-risks development decisions in biologics and nucleic acids projects. We are excited to expand our network in the West Coast".
Alex Chen, CEO of Crystal Pharmatech
Event Details
- Date: February 24, 2026
- Time: 5:30 PM to 8:30 PM PST
- Location: San Diego, Sorrento Mesa Area, California
- Schedule:
- 5:30 PM Opening remarks
- 5:45 PM Networking and cocktails
- 8:15 PM Closing remarks
Attendance is free of charge. This is an open gathering for the San Diego biotech community.
For logistical information, contact: bd_global@crystalpharmatech.com
Who You Will Meet
Scientific and technical leaders from Crystal NAX will be present to discuss nucleic acid and LNP development topicsLeaders from the broader Crystal organizations (including Crystal Bio Solutions and Crystal Pharmatech) will also be present to support cross-modality conversations.
Discussion topics may include:
- IND-enabling CMC strategy for RNA modalities
- RNA impurity risk and analytical control (including dsRNA control approaches)
- LNP formulation development, scale-up considerations, and process sensitivity
- Stability strategy and comparability readiness for platform changes
- Practical analytics that support release, stability, and mechanistic understanding
Questions & Answers
Is registration required for the San Diego biotech networking event? Yes. Please register through the event page to confirm attendance. The reception is open to biotech and pharma R&D professionals.
Is this event only for RNA teams? No. While Crystal NAX focuses on RNA and LNP development, colleagues from Crystal Pharmatech (CPT) and Crystal Bio Solutions (CBS) will also attend. CPT supports small molecule development, and CBS focuses on biologics characterization, CMC analytics, and bioanalysis.
Who should attend from a biotech company? CMC leads, analytical development scientists, RNA and formulation teams, bioanalytical scientists, translational and clinical development leads, and founders or technical decision-makers evaluating development and manufacturing partners across small molecules, biologics, and nucleic acid programs.
Can we discuss IND-enabling CMC strategy? Yes. Informal discussion about analytical readiness, comparability risk, and control strategy is appropriate. Formal regulatory advice requires structured engagement.
Is this a technical seminar or presentation? No. The format is conversational.
Can we explore potential collaboration? Yes. The reception supports early-stage discussion about fit and technical approach before formal vendor engagement.